Recommendations from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®)1
- IDH1 mutations have been reported in approximately 10% to 20% of intrahepatic CCA cases. Activating IDH1 mutations can lead to high levels of 2-HG accumulation and impact normal liver cellsa
- “Testing for IDH1 mutations is recommended in patients with unresectable or metastatic intrahepatic CCA or extrahepatic CCA"
- Ivosidenib (TIBSOVO) is recommended as an NCCN category 1 subsequent-line systemic treatment option for patients with unresectable or metastatic progressive CCA with IDH1 mutationsb
2-HG, 2-hydroxyglutarate; CCA, cholangiocarcinoma; NCCN, National Comprehensive Cancer Network® (NCCN®).
aImpairment of normal differentiation, accumulation of hepatic progenitor cells, and malignant transformation to intrahepatic CCA.1
bTo view the most recent and complete version of the guidelines, visit NCCN.org.
IDH1 mutations are common genetic alterations in cholangiocarcinoma
mIDH1 is an early driver mutation that occurs in up to 20% of intrahepatic cholangiocarcinoma cases in the United States2,3Biomarker testing for mIDH1 can be performed by next generation sequencing of tumor tissue using an FDA-approved companion diagnostic4
TIBSOVO® (ivosidenib tablets) selectively inhibits mIDH1 and may play an important role in filling an unmet need in the treatment of advanced cholangiocarcinoma4,5
IDH1, isocitrate dehydrogenase-1; mIDH1, mutated IDH1.
IDH1 mutations and cholangiocarcinoma
IDH1 mutations can play a critical role in the development of cholangiocarcinomaIDH1 mutations confer gain-of-function activity6
- Isocitrate dehydrogenase (IDH) is a key enzyme in the citric acid cycle that catalyzes the conversion of isocitrate to α-ketoglutarate (α-KG)6-8
- Cancer-associated mutations in IDH1 catalyze the reduction of α-KG to the oncometabolite 2-hydroxyglutarate (2‑HG)6,8
- Accumulation of 2-HG leads to oncogenesis8
- Elevated levels of 2-HG have been observed across multiple tumors with IDH1/2 mutations, including acute myeloid leukemia, glioma, and cholangiocarcinoma7,8
Cholangiocarcinoma incidence and survival
Incidence of intrahepatic cholangiocarcinoma is increasing in the United States9
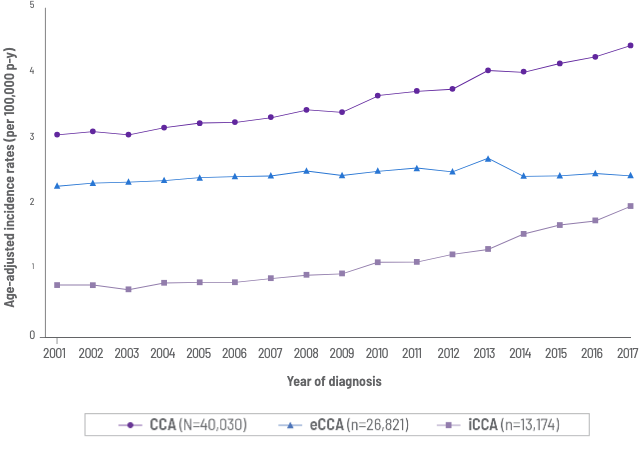
- Cholangiocarcinoma is the second most common primary liver cancer and is associated with a poor prognosis9
- Overall age-adjusted incidence: 3.65 per 100,000 (p-y)9
- Incidence of intrahepatic cholangiocarcinoma increased 148.8% (0.80 to 1.99) from 2001 to 20179
- Most patients (67%) are ≥65 years at diagnosis9
- Largely due to a lack of specific symptoms, most patients (~70%) are diagnosed at late stages when the disease is unresectable9
Prognosis For Cholangiocarcinoma
The 5-year relative survival rate for metastatic intrahepatic cholangiocarcinoma is 3%10,c
P-y, person-years.
cFor patients whose initial diagnoses were given after their cancer had already metastasized.10


