The ClarIDHy trial for TIBSOVO was the first phase 3 trial to show clinical benefit with targeted therapy in mIDH1 CCA and one of the largest to date1-3
The primary endpoint was progression-free survival (PFS), and secondary endpoints included overall survival, objective response rate, safety, and quality of life.1,2
TIBSOVO was studied in a phase 3, randomized, double-blind, placebo-controlled, multicenter trial in a CCA patient population reflective of that seen in clinical practice.1-3
Recommendation from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®)4
Ivosidenib (TIBSOVO) is recommended as an NCCN category 1 subsequent-line systemic treatment option for patients with unresectable or metastatic progressive CCA with IDH1 mutations.4,a
aTo view the most recent and complete version of the guidelines, visit NCCN.org.
mIDH1, mutated isocitrate dehydrogenase-1; NCCN, National Comprehensive Cancer Network® (NCCN®).
TIBSOVO delivered significant improvements in PFS1
PFS for TIBSOVO vs placebo1,2
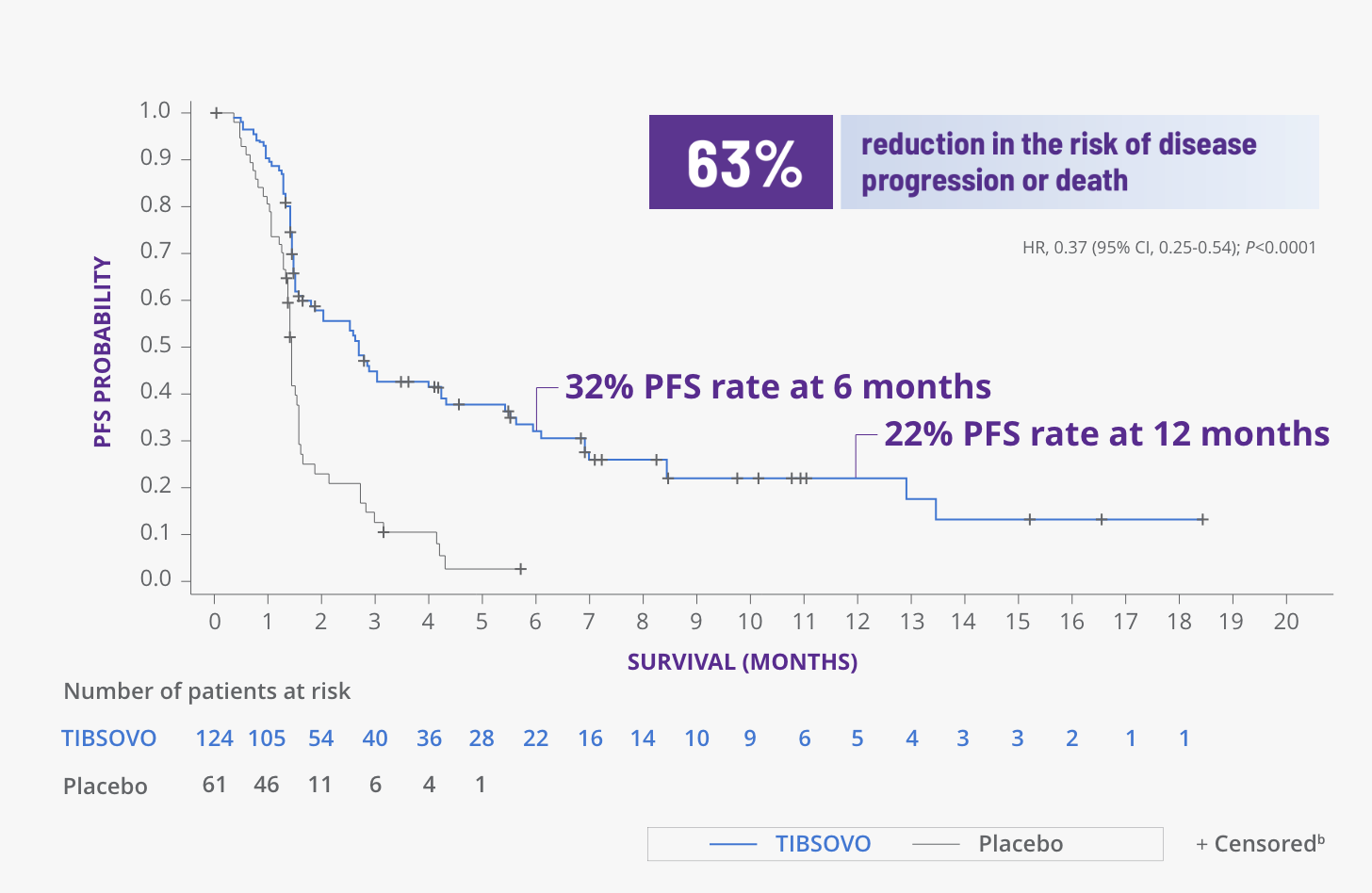
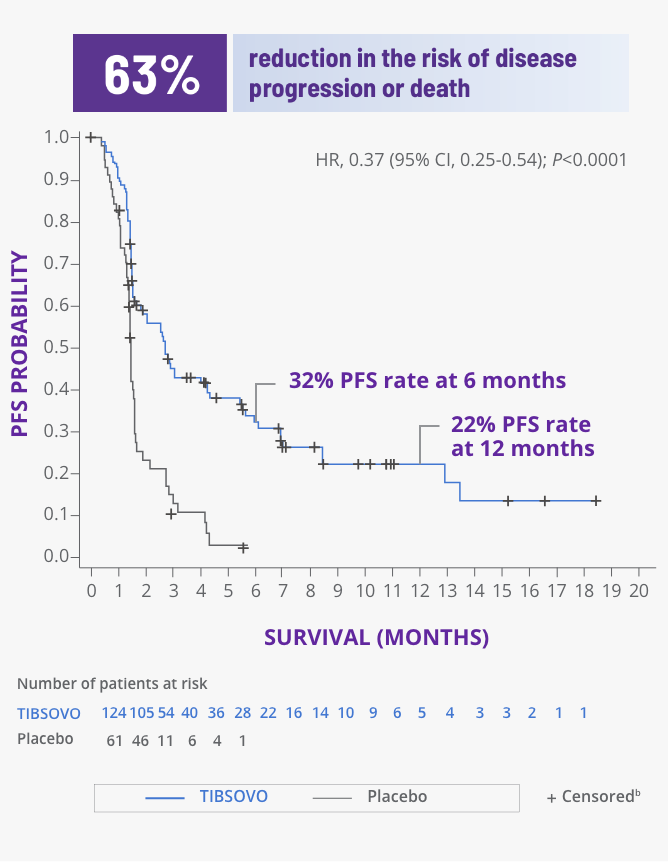
CI, confidence interval; HR, hazard ratio.
bPatients were censored if they were missing a baseline assessment, missing documented progression or death before data cutoff, started alternate anticancer systemic treatment, or had documented progression or death following 95 days or more from the previous adequate assessment.2
- None of the patients assigned to placebo had PFS of 6 months or greater2
- The median PFS was nearly twice as long for patients assigned to TIBSOVO compared with placebo (2.7 months [95% CI, 1.6-4.2] versus 1.4 months [95% CI, 1.4-1.6])2
Treatment duration within ClarIDHy trial
MEDIAN TREATMENT DURATION
2.8 months
with TIBSOVO1
(range, 0.1 to 34.4 months)
1.6 months
with placebo2
(range, 0 to 6.9 months)
Treatment duration for all patients treated with TIBSOVO5

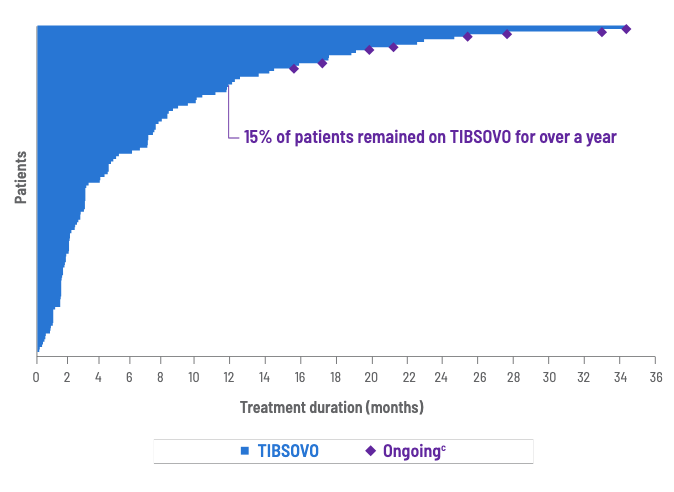
cOngoing as of data cutoff (May 31, 2020).
Treatment duration for all patients treated with placebo, including those who crossed over to TIBSOVO5

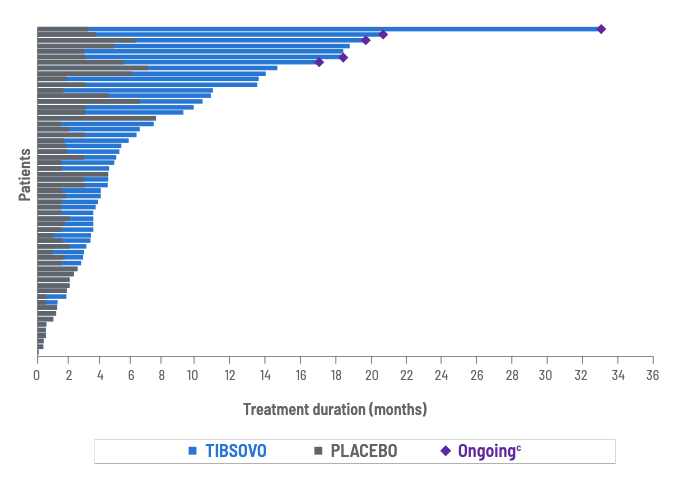
cOngoing as of data cutoff (May 31, 2020).
Overall survival (OS) data with TIBSOVO
OS for TIBSOVO vs placebo (intent-to-treat set)5,d
|
TIBSOVO n=126 |
Placebo n=61 |
Placebo (adjusted for crossover) n=61 |
|
|---|---|---|---|
| Number of events (%) | 100 (79.4) | 50 (82.0) | 49 (80.3) |
| Median OS, months | 10.3 | 7.5 | 5.1 |
| 12-month rate | 43% (95% CI, 34%-51%) |
36% (95% CI, 24%-48%) |
36% (95% CI, 24%-48%) |
CI, confidence interval; HR, hazard ratio; RPSFT, rank-preserving structural failure time.
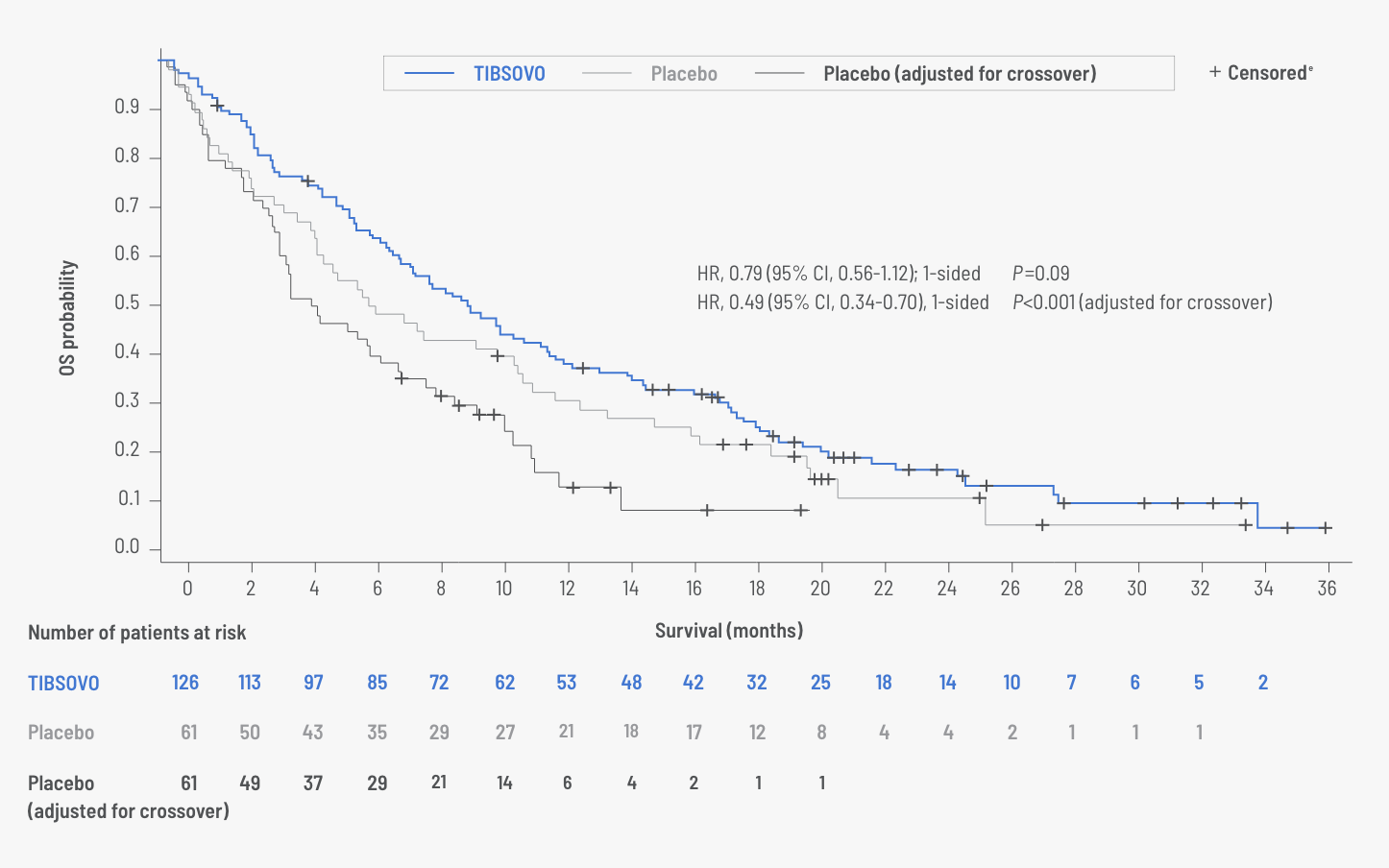
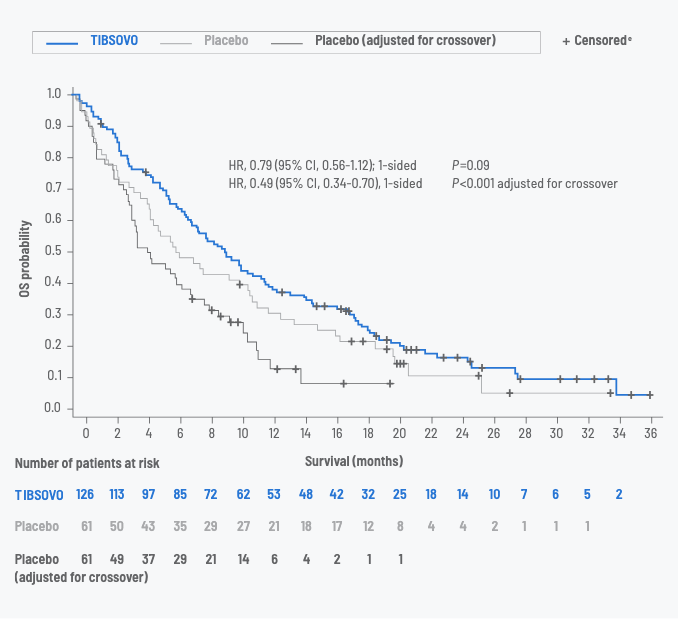
dOS results are based on the final analysis of OS (based on 150 deaths), which occurred 16 months after the final analysis of PFS.1
ePatients without documentation of death at the time of the data cutoff for analysis were censored at the date the subject was last known to be alive or the data cutoff date, whichever was earlier.2
- Among the 61 patients in the placebo arm, 70% crossed over to receive TIBSOVO1
- These patients continued to be grouped within their original randomized assignment for analysis purposes3
- As a result, OS data may be confounded by the crossover2
- A statistical model known as rank-preserving structural failure time (RPSFT) was used to recreate the placebo curve as if crossover had not occurred6,f,g
- When adjusted for crossover, median OS for the placebo group was 5.1 months (95% CI, 3.8-7.6 months; HR, 0.49 [95% CI, 0.34-0.70])5
fThis prespecified model, known as RPSFT, is one of the most commonly used statistical methods to adjust for treatment switching in oncology trials. This sensitivity analysis was exploratory, and appropriate multiplicity adjustments were not applied.6
gThe RPSFT model is based on the assumption that the treatment effect of TIBSOVO is the same for all individuals, regardless of when treatment is received. This assumption was not verified.5
More than half (51%) of patients receiving TIBSOVO achieved stable disease5
Disease control rate5,h
53%
with TIBSOVO
28%
with placebo
ORR, objective response rate; SD, stable disease.
hThe disease control rate (ORR + SD) was due mostly to the SD rate (an ORR rate of 2% [3 partial responses] and an SD rate of 51% with TIBSOVO vs an ORR of 0% and an SD rate of 28% with placebo).
Health-related quality of life (QoL)
- CCA is associated with an impaired QoL, a metric that is increasingly regarded as a crucial endpoint in clinical trials7
- Health-related QoL was assessed using two tools; the EORTC QLQ-C30, a generic cancer questionnaire, and the EORTC QLQ-BIL21, a disease-specific module5
- TIBSOVO was favored on physical functioning, as well as subscales such as pain, emotional functioning, cognitive functioning, dyspnea, anxiety, and tiredness5
- Analyses of the QoL data were limited by small sample sizes.5 Assessments depicted were at Cycle 2 Day 15,i
EORTC QLC-C30 global health status/QoL and functional subscales8


EORTC QLQ-C30 symptoms subscales8

EORTC QLQ-BIL21 subscales8

These QoL analyses were exploratory, and appropriate multiplicity adjustments were not applied. Results of these individual components need cautious interpretation and could represent chance findings.
CI, confidence interval; EORTC QLQ-BIL21, EORTC QLQ cholangiocarcinoma and gallbladder cancer module; EORTC QLC-C30, European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30; LS, least squares.
i At baseline: n=53 for placebo and n=114 for TIBSOVO (QLQ-C30); n=52 for placebo and n=108 for TIBSOVO (QLQ-BIL21). At Cycle 2 Day 1: n=24 for placebo and n=68 for TIBSOVO (QLQ-C30); n=23 for placebo and n=68 for TIBSOVO (QLQ-BIL21). At Cycle 3 Day 1: n=12 for placebo and n=52 for TIBSOVO (QLQ-C30); n=12 for placebo and n=52 for TIBSOVO (QLQ-BIL21).5


